HS Dosing COSENTYX® (secukinumab)
4.9 (117) · € 11.50 · En stock
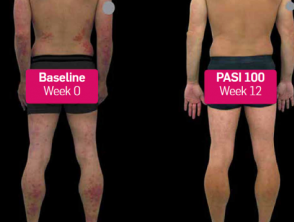
Secukinumab

Novartis' Cosentyx gets US nod for hidradenitis suppurativa

Full article: Biologic Treatments of Psoriasis: An Update for the Clinician
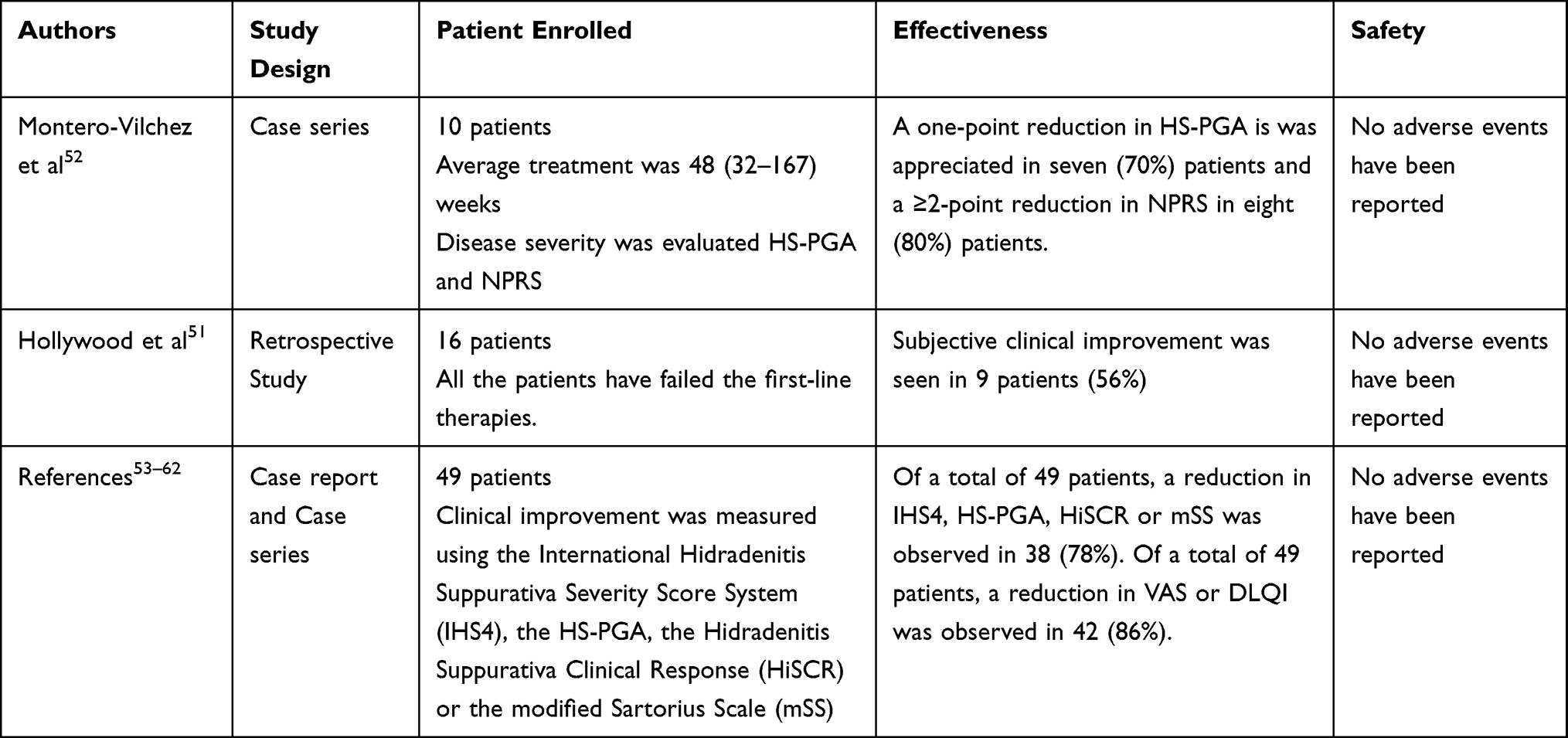
Management of Hidradenitis Suppurativa

Cosentyx - NPS MedicineWise

US approval for fourth Cosentyx indication

Secukinumab - Wikipedia
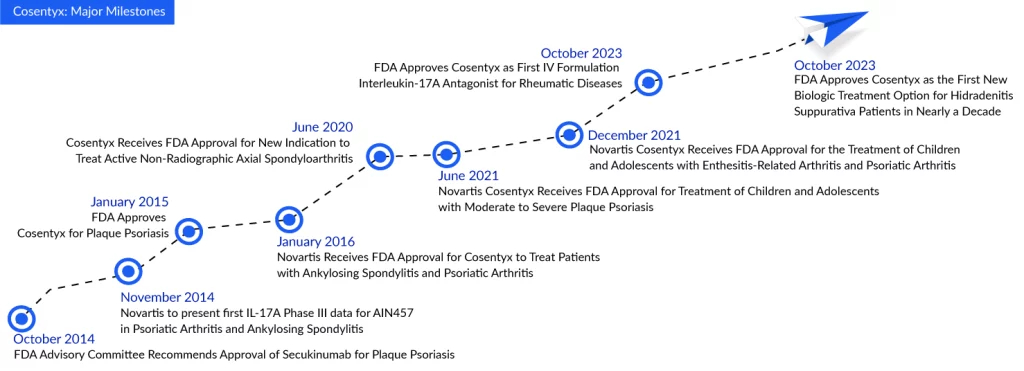
Cosentyx: First Biologic Hidradenitis Suppurativa Treatment
Cosentyx: Package Insert
Systematic review of immunomodulatory therapies for hidradenitis suppurativa. - Document - Gale OneFile: Health and Medicine

Secukinumab for Patients with Moderate-to-Severe Hidradenitis Suppurativa: Post Hoc Analyses of Efficacy Endpoints, Draining Tunnels, Skin Pain, and Work Productivity - European Medical Journal

Cosentyx Dosage Guide

Full article: Real-world dose escalation of biologics for moderate-to-severe psoriasis in the United States

Office Resources, COSENTYX® (secukinumab)

Delayed drug hypersensitivity reaction to secukinumab in a patient with hidradenitis suppurativa












